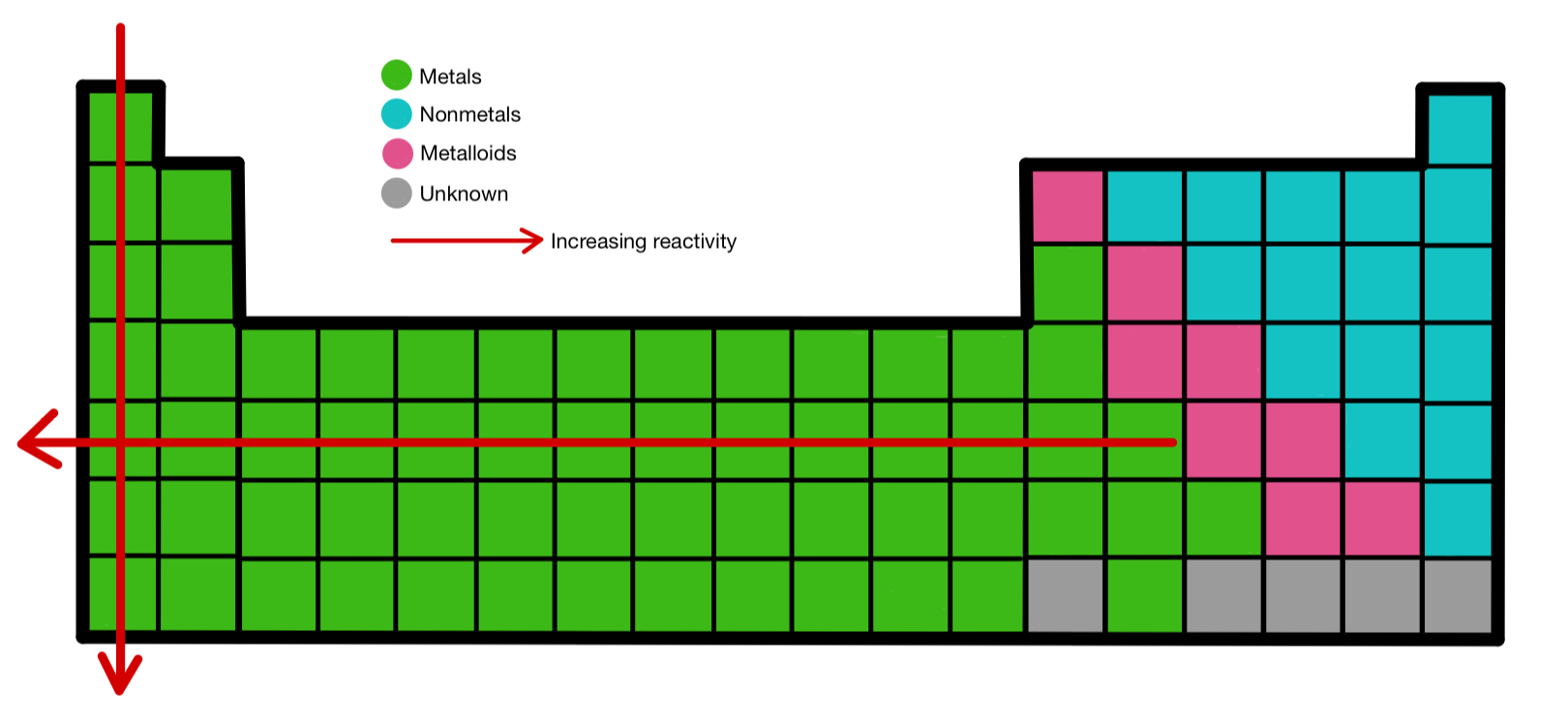
In fact, it’s an element of extremes with a wide range of uses, and it could surprise us in the future.įluorine (atomic symbol: F) is the lightest member of the halogen group, commonly known as group 17 in the Periodic System of Elements, and the 13th most prevalent element in the earth’s crust.įluorine is a diatomic gas with a yellowish hue under normal conditions. Cesium is the most reactive element since it is the second from the bottom of this group, has six electron shells, and exhibits the characteristics of a reactive atom.įluorine is a noteworthy chemical element for many reasons, not the least of which is its unusual reactivity. The most reactive substance – Alkali metals are the most reactive element group (situated far apart from intermediate metals and noble gasses). The ability of metals to donate electrons decreases as they progress through the activity series, which is an essential property. It can also be used to determine metal reactivity in the presence of water and acids. The reactivity series data can be used to determine if a metal can displace another in a single displacement reaction. These metals discolor and corrode quickly. 
Because they are easily oxidized, the metals near the top of the reactivity range are powerful reducing agents. The activity series, sometimes known as the reactivity series, is a list of metals arranged in descending order of their activities. Let’s start with the most reactive element and the reactivity series. The reactivity series is used to determine an element’s reactivity. 
Answer: Cesium is the most reactive element since it is the second from the bottom of this group, has six electron shells, and exhibits the characteristics of a reactive atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
